National Advisory Committee on Immunization (NACI) is now recommending a booster dose of COVID-19 vaccine at least 6 months after completion of a primary COVID-19 vaccine series for all Canadians aged 18 and above.
The updated guidance comes as new COVID-19 cases are rising across Canada with Ontario reporting 1000+ new cases for the first time in 6 months.
NACI says the recent evidence suggests that vaccine effectiveness against SARS-CoV-2 virus that causes COVID-19 is decreasing over time. NACI expects booster doses of COVID-19 mRNA vaccines to increase the immune response and offer enhanced protection against infection, severe disease and may help reduce the spread of infection.
“NACI has continued to closely monitor the evidence on the benefit and need for booster doses in Canada, and is now providing updated advice on the use of booster COVID-19 vaccine doses. Although protection against severe disease remains high in the general population, we know that protection against infection can decrease over time. We have also seen an increase in the risk of severe disease for select groups who are vaccinated, particularly older adults, ” said NACI Chair Dr. Shelley Deeks in a news release.
“Offering booster doses will help ensure that protection against severe disease remains high, and may have an impact on spread in the community as well. It is important to note that there is no information yet on the impact of the new variant, Omicron, on the effectiveness of the vaccine. NACI will continue to monitor and provide updated and timely advice as necessary.”
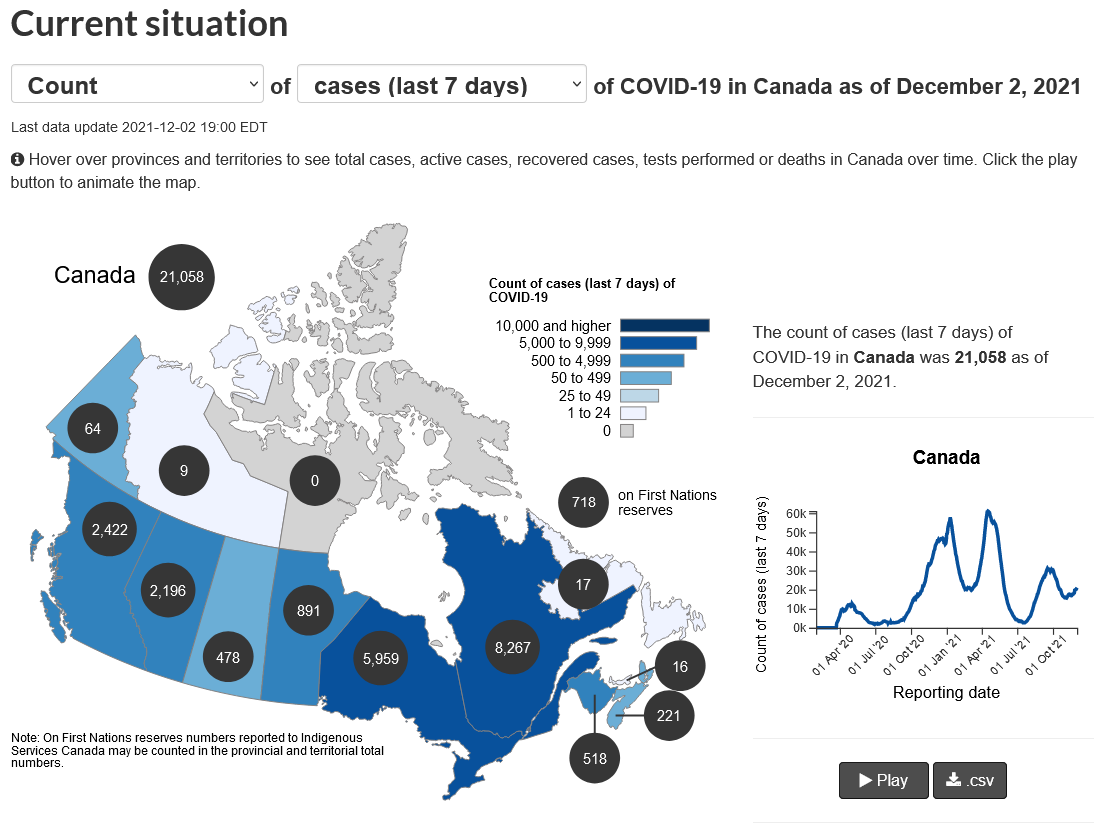
New Cases in the last days across Canada. Credit: Canada.ca
NACI is recommending Pfizer-BioNTech Comirnaty 30 mcg vaccine for a booster dose in those 18 to 29 years of age due to the lower risk of myocarditis/pericarditis with this vaccine over Moderna Spikevax booster dose (50 mcg dose).
According to the news release, in Israel, Myocarditis and/or pericarditis after mRNA vaccination remain rare and the rates of myocarditis and/or pericarditis have been lower than what was observed with the second dose of the primary series, but higher than what was observed with the first dose.
In people over 30 years of age, either mRNA vaccine can be used as the booster dose.
The recommendations also include moderately to severely immunocompromised adults who have received a three- dose primary COVID-19 vaccine series. NACI says though the data on the fourth dose of a COVID-19 vaccine after the recommended three-dose primary series in this population is currently limited, these individuals may also receive a booster at least 6 months after receiving their last dose.
NACI says if Moderna is used as a booster dose for moderately to severely immunocompromised, and adults living in long-term care for seniors or other senior congregate living settings, then 100 mcg Moderna Spikevax is preferred.
Updates booster recommendations by National Advisory Committee on Immunization :
- NACI strongly recommends a booster dose of an authorized mRNA COVID-19 vaccine should be offered at least 6 months after completion of a primary COVID-19 vaccine series to the following groups:
o People aged 50 years and older
o Adults living in long-term care homes for seniors or other congregate living settings that provide care for seniors
o Recipients of a viral vector vaccine series completed with only viral vector vaccines (AstraZeneca/COVISHIELD or Janssen COVID-19 vaccine)
o Adults in or from First Nations, Inuit and Métis communities
o All frontline healthcare workers having direct in-person contact with patients - NACI recommends that a booster dose of an authorized mRNA COVID-19 vaccine may be offered to adults 18 to 49 years of age at least 6 months after completion of a primary COVID-19 vaccine series with consideration of jurisdictional and individual risks as outlined here.
The committee said they are studying the new COVID-19 variant of concern, Omicron.
You may like:







