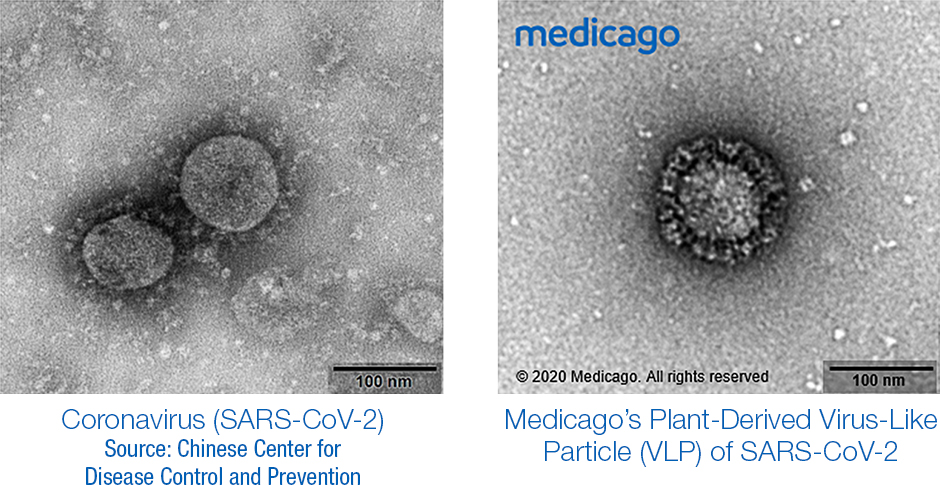
Credit: Medicago
Medicago, a Quebec biopharmaceutical company says the company has started Phase 1 clinical trials of COVID-19 vaccine.
Drugmakers are racing to make a successful vaccine; with different groups working on different technology and methodology for a collective solution to COVID-19. Medicago’s vaccine is plant-based and its development is essential in developing a diverse range of COVID-19 vaccines.
As part of its Phase 1 trial, the first doses of the vaccine candidate were administered to healthy human volunteers on Monday. This is the first vaccine from Canada to start human trials. During the trial, random 180 healthy male and female candidates, aged 18 to 55, would be dosed with the vaccine candidate alone and with two different adjuvants – from GlaxoSmithKline and Dynavax Technologies Corp. The use of the adjuvants is thought to boost immune response, thereby allowing the production of more vaccine doses.
As per the news release, pre-clinical results of the vaccine candidate with and without the booster were very encouraging. Should the Phase 1 trial prove to be successful, Medicago plans to enter Phase 2/3 trials in October. Medicago expects to complete development and manufacture 100 million doses by the end of 2021, contingent on clinical development and regulatory considerations. This will be increased to 1 billion doses annually on completion of its large-scale facility in Quebec City by 2023.
Medicago’s approach takes the leaves of a plant as a bioreactor to produce one of the three spike proteins of the novel coronavirus, the S-spike. The plant is a close relative to tabacco, known as Nicotiana Benthamiana. The plant has a weak immune system which makes it a good host for virus-like particles (VLP). The company is also using this technology to develop antibodies against SARS-CoV-2.
- Alberta Company Plans to Start Made-in-Alberta COVID-19 Vaccine Human Clinical Trials by Late July
- First Clinical Trial For COVID-19 Vaccine Approved By Health Canada







