
Credit: Ian Hutchinson
Health Canada has authorized the use of the Moderna COVID-19 vaccine for children aged 12 and up in Canada.
Until now, only Pfizer’s COVID-19 vaccine for available for children between the ages of 12 and 17.
(1/3) Health Canada has authorized the use of the Moderna #CovidVaccine in youth aged 12 to 17. Until now, this vaccine was only authorized for use in people 18 years of age or older. pic.twitter.com/xMqf6MGR0A
— Health Canada and PHAC (@GovCanHealth) August 27, 2021
Announcing the approval, Health Canada said, “After a thorough and independent scientific review of the evidence, Health Canada has determined that the vaccine is safe and effective at preventing #Covid19 in youth aged 12 to 17.”
Statement from the Chief Public Health Officer of Canada reads, “In its review of the evidence for this updated guidance, NACI considered the rare cases of myocarditis (inflammation of the heart muscle) and/or pericarditis (inflammation of the tissue surrounding the heart) following immunization with mRNA COVID-19 vaccines, reported both in Canada and internationally. Although, a higher rate of myocarditis and/or pericarditis adverse events were recently reported after administration of the Moderna COVID-19 vaccine compared to the Pfizer-BioNTech vaccine, these events are still considered rare and verification of this potential difference is ongoing. Nevertheless, some provinces and territories may decide to continue using the Pfizer-BioNTech given more experience with this vaccine product in the adolescent age group and the possibility of a lower rate of myocarditis and/or pericarditis. ”
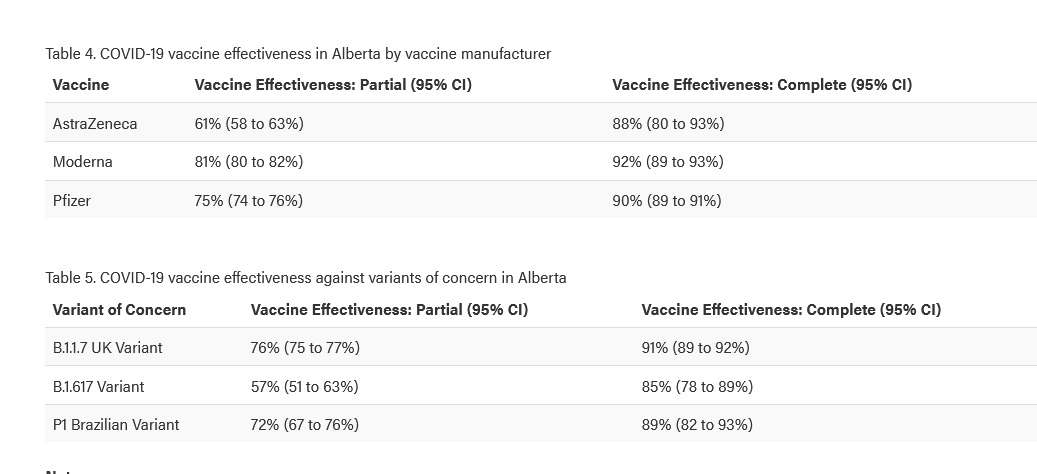
As per Alberta Health data, the Moderna vaccine is 92% effective in preventing symptomatic infection due to COVID-19, while Pfizer vaccine is 90% effective.
Credit: Alberta.ca
Recently, Moderna signed a memorandum of understanding (MOU) to build a state-of-the-art mRNA vaccine production facility in Canada.







