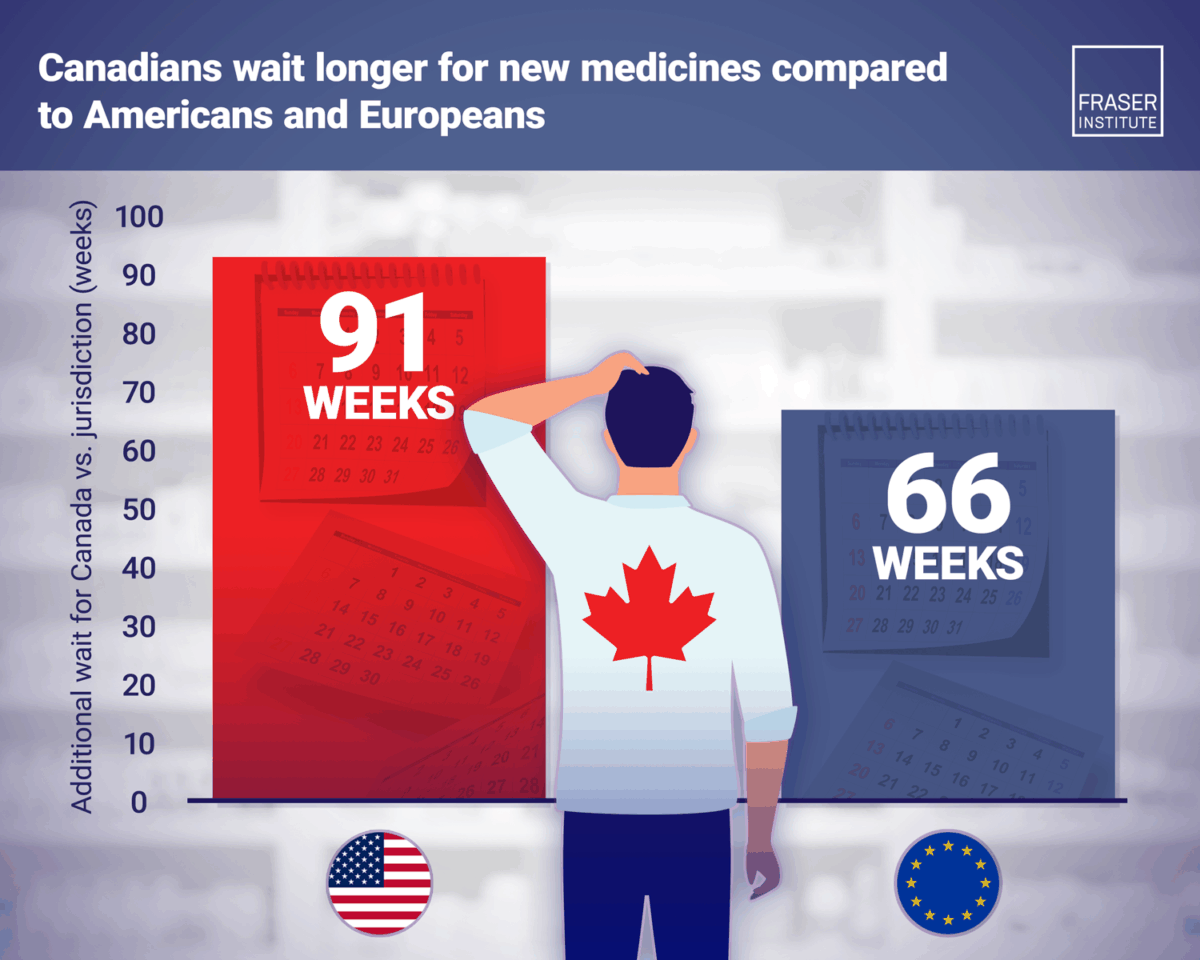
Canadians are waiting significantly longer for access to new medicines compared with patients in other developed countries, according to a new study released by the Fraser Institute.
The research finds delays average more than 65 weeks compared with Europe and over 90 weeks compared with the United States.
The study, Timely Access to New Pharmaceuticals in Canada, the United States, and the European Union (2026), was authored by Kristina Acri, a senior fellow at the institute. It examines how quickly new medicines move through approval systems in Canada, the U.S., and the European Union.
Between 2019/20 and 2024/25, 194 drugs were approved in both Canada and the United States. On average, approval in the U.S. came 636 days earlier, with a median gap of 371 days. During the same period, 174 drugs approved in both Canada and Europe received authorization 459 days earlier in Europe, with a median difference of 211 days.
Acri points to drug submission timing as the primary cause. Pharmaceutical companies often delay submitting applications to Health Canada, sometimes by up to a year after approvals are granted by the US Food and Drug Administration or the European Medicines Agency.
The study suggests this hesitation reflects broader concerns, including Canada’s smaller market, weaker intellectual property protections, and strict federal pricing policies. As a result, Canada’s approval process often duplicates reviews already completed elsewhere, extending wait times for patients.
- Health Care Wait Times in Canada 2025: How Long You Need to Wait For Medical Treatment
- Emergency Rooms Wait Times: How Long You Have to Wait in Emergency Rooms Across Canada
According to the report, these delays are not just administrative. They are linked to fewer treatment options, poorer health outcomes, and higher overall health-care costs. Acri argues that without international recognition of approvals, policymakers should reassess how current drug policies affect Canada’s ability to attract new medicines.
The study concludes that Canadians would see faster access to treatments under approval systems similar to those used in the U.S. or Europe.